|
This breaks off the blue section, like a backbone on the molecule, which becomes glycerol. Doing this reaction requires using methanol (shown in green), which causes the red bonds in the structure below to break. Therefore, converting vegetable oil into biodiesel is called a transesterification reaction. Vegetable oil, like biodiesel, belongs to a category of compounds called esters. Weve seen that carboxylic acid derivatives react with nucleophiles to give substitution. 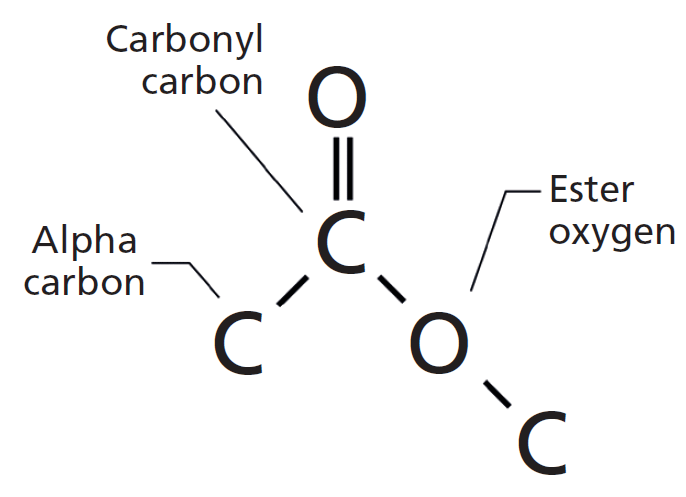
Chemical Conversion of Vegetable Oil to Biodiesel Well also examine the chemistry of the ester functional group in fats. Converting it into biodiesel makes it a smaller molecule, closer to the size of regular diesel, so that it has to get colder than vegetable oil before it starts to gel. That larger size of vegetable oil means that in cold weather it gels, making it hard to use in an engine. It also has ester functional groups (in blue), like biodiesel. Notice that it also has the long rows of carbon and hydrogen atoms, but is about three times larger than normal diesel molecules. Notice that regular diesel also has the long chain of carbon and hydrogen atoms, but doesn’t have the ester group shown in blue above.Īctually, the first diesel engines didn’t run on “diesel” fuel, but on vegetable oil, a sample molecule of which is shown below. This is possible because biodiesel is chemically very similar to regular diesel, shown below. Mostly it is a long chain of carbon atoms, with hydrogen atoms attached, and at one end is what we call an ester functional group (shown in blue).ĭiesel engines can burn biodiesel fuel with no modifications (except for replacing some rubber tubing that may soften with biodiesel). The good is that esters follow the same pattern and instead of the metal ion, we use the alkyl group connected to. For example, sodium acetate, potassium butyrate, etc. Differences Between Biodiesel, Diesel and Vegetable OilĪ typical molecule of biodiesel looks like the structure below. In the IUPAC nomenclature of carboxylic acids, we learned that their salts are named by replacing the suffix ic acid or oic acid with ate. In a solution of a strong acid, the molecules are fully ionised, but in a weak acid, very few of the molecules are ionised. The pH of a weak acid will be higher than the pH of a strong acid, if their concentrations are the same. An indispensible resource for the organic chemist, this. This means that their solutions do not contain many hydrogen ions compared with a solution of a strong acid with the same concentration. Patais Chemistry of Functional Groups is one of chemistrys landmark book series in organic chemistry. The general equation for the formation of an ester is:Īlcohol + carboxylic acid → ester + waterĮthanol + ethanoic acid → ethyl ethanoate + water Weak and strong acids - HigherĬarboxylic acids are weak acids. The center of the ester functional group is the carbon double bond oxygen. The two carbons with all single bonds is the center of a tetrahedral geometry. Chime: Ester : There are several centers of interest. Esters have fruity smells and can be used as solvents. An additional molecular geometry is centered on the oxygen of the - OH group. Esters are organic compounds which all contain the functional group -COO. Making estersĬarboxylic acids can react with alcohols to make esters. These properties are due to the –COOH functional group. react with carbonates to form a salt, water and carbon dioxide.react with bases to form a salt and water.react with metals to form a salt and hydrogen.dissolve in water to form acidic solutions with pH values less than 7.The carboxylic acids have the typical properties of acids. The table shows four carboxylic acids, their molecular formulae and their structures. Vinegar is a dilute solution of ethanoic acid. In an electrolytic cell, oxidation occurs at the cathode and reductions at the anode D. when a redox reaction reaches equilibrium. For a spontaneous redox reaction, the reaction potential E is negative B. 
Which of the following statements is true A. It is responsible for the typical reactions of carboxylic acids, which are weak acids. which one of these structures represents an ester functional group 0 18. The functional group in the carboxylic acids is the carboxyl group, -COOH. show a gradual variation in physical properties, such as their boiling points. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
